Bio-Rad Laboratories announced the launch of new rapid cell lysis kits that allow researchers to obtain reverse transcription quantitative PCR (RT-qPCR) data directly from cultured cells without the need for a separate RNA purification step. Bio-Rad’s SingleShot™ family of cell lysis RT-qPCR kits provide high-quality gene expression results in less than two hours.
Available column isolation methods for purifying RNA are time-consuming and laborious while other methods that enable RT-qPCR directly from cell lysates can damage the RNA and result in poor genomic DNA clearance. However, Bio-Rad’s SingleShot Kits eliminate those challenges and offer superior reproducibility and accuracy of gene expression results. In addition, minimal setup and pipetting steps create an automation-friendly workflow and, unlike other similar methods, Bio-Rad’s kits do not require an additional pipetting step to stop the cell lysis reaction.
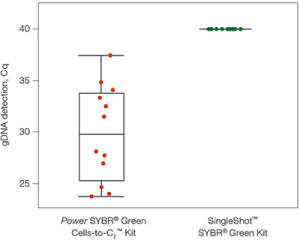
The SingleShot Kit removed genomic DNA (Cq > 40) while another available kit resulted in poor genomic DNA clearance.
“The SingleShot Kits are ideally suited for high-throughput laboratories with large-volume workloads and for researchers who are faced with a limited number of cells and require extreme accuracy in each analysis,” said Paul Streng, senior product manager in the Gene Expression Division of Bio-Rad’s Life Science Group.
Bio-Rad’s SingleShot Kits are the only available kits that include an RNA control template and qPCR assay to help researchers determine optimal cell number and lysate inputs for their RT-qPCR reactions.
SingleShot Kits are validated for use with a wide variety of adherent and suspension cell lines and are offered in multiple formats, including one-step RT-qPCR and two-step RT-qPCR kits that are compatible with either SYBR® Green or probe-based assays. SingleShot Kits are available as a stand-alone cell lysis kit. All SingleShot Kits are validated for use with PrimePCR™ Assays and Panels.

















 Bio-Rad Laboratories announced the launch of the
Bio-Rad Laboratories announced the launch of the  Bio-Rad Laboratories is sponsoring a new version of the
Bio-Rad Laboratories is sponsoring a new version of the